A within-person analysis of magnesium's effect on sleep at consumer-wearable scale
Users log a +1.9-point higher sleep score and a 0.8 bpm lower overnight heart rate on nights they took magnesium versus their own non-magnesium nights. 71 % of members show a positive within-user sleep-score difference on their magnesium nights.
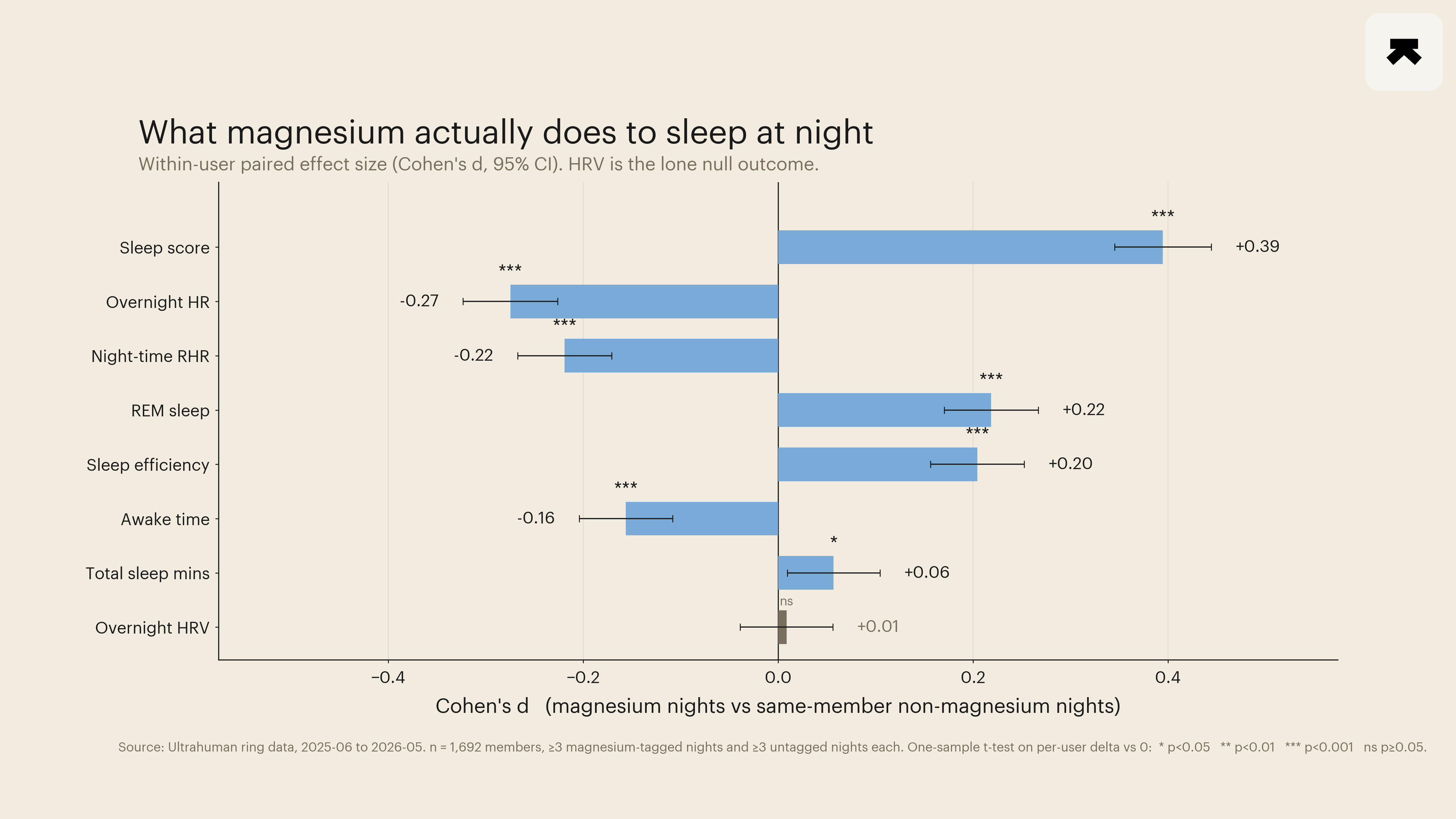
The clinical literature on magnesium and sleep has been stuck in the same place for most of the last decade: a handful of placebo-controlled trials, none of them larger than 160 participants, with effect sizes small enough that meta-analytic pooling has been unable to deliver a confident summary statement. A within-person paired analysis of 1,692 Ultrahuman members across 337,471 ring-recorded nights provides one piece the literature has been missing. On the same person's magnesium-tagged nights, sleep score rose by 1.88 points (95 % CI 1.65 to 2.10, Cohen's d 0.39, Holm-corrected p ≈ 2 × 10⁻⁵⁸), and overnight heart rate dropped by 0.81 bpm (CI 0.95 to 0.67, d 0.27). Six of the eight primary outcomes moved in the favourable direction at the same significance level. Overnight heart-rate variability was the lone null outcome.
The most rigorous recent randomised controlled trial of magnesium for sleep is by Schuster et al. (2025), a 4-week double-blind trial of 250 mg elemental magnesium bisglycinate in 155 adults reporting poor sleep quality. The trial reported a statistically significant but modest reduction in Insomnia Severity Index scores in the magnesium arm (Cohen's d=0.2). A separate 21-day trial of magnesium L-threonate (Hausenblas et al., 2024) used wearable ring instrumentation alongside subjective questionnaires and reported objective gains in deep and REM sleep scores, distinguishing it as the only recent trial in the literature with a wearable-derived outcome. A 2021 systematic review and meta-analysis (Chan and Lo, 2021) had to handle magnesium narratively rather than meta-analytically because the pooled trial-level data were sparse and heterogeneous in form, dose, and outcome instrument. A small RCT of zinc-magnesium-aspartate in trained men under partial sleep deprivation (Gallagher et al., 2024) found no effect on subsequent morning performance or subjective sleep, providing a useful null-leaning counterweight in a different cohort and combination.
The mechanistic plausibility is well-rehearsed (Yeom and Cho, 2024): magnesium is a positive allosteric modulator of GABA-A receptor function, an antagonist of NMDA-type glutamate receptors at physiological intra-neuronal concentrations, and a co-factor in the methylation step that produces melatonin from N-acetylserotonin.
None of these pathways predict a large pharmacological effect at supplemental doses, but each predicts a small, dose-modulated tilt in autonomic and circadian function consistent with what the recent placebo-controlled trials have been finding.
We extracted every smart-tag log keyed to one of three magnesium variants (generic magnesium, magnesium bisglycinate, magnesium L-threonate) recorded in the Ultrahuman application between 2025-06-04 and 2026-05-05. Tags were attributed to the night of the logged date. Members were retained if they logged at least three magnesium-tagged nights and contributed at least three untagged nights with a valid ring-recorded sleep score in the same window. The eligible cohort comprised 1,692 members, contributing 14,916 magnesium nights and 322,555 untagged nights. The analysis design is a paired within-person test: for each member, condition-conditional means were computed across the eligible nights, and a one-sample t-test on the per-member differences was applied to each outcome with Holm-Bonferroni correction across the eight primary contrasts.
Six outcomes crossed the significance threshold at the cohort level. Sleep score rose by 1.88 points (Cohen's d=0.39). Average overnight heart rate fell by 0.81 bpm (d=0.27). The heart-rate signal was reproduced on a separate channel: night-time resting heart rate, computed during the sleep window only, fell by 0.60 bpm (d=0.22). REM sleep duration rose by 4.06 minutes (d=0.22). Sleep efficiency rose by 0.62 percentage points (d=0.20). Awake time fell by 2.66 minutes (d=0.16). Total sleep duration moved by approximately two and a half minutes (p=0.02 unadjusted, p 0.04 Holm), too small to interpret as a clinically meaningful shift in time asleep. Overnight heart-rate variability, computed as the per-night ring-derived RMSSD-equivalent, did not move (mean within-person difference=0.05 ring units, CI crossing zero, p=0.72). The cardiac quieting on tagged nights is therefore on the rate side, not the variability side, a separation that small clinical trials have generally been underpowered to characterise.
Two stratifications are notable. Members who logged magnesium on ten or more nights in the window (N=347) showed the largest effect on sleep score (d=0.52), nearly half a standard deviation. The pattern is consistent with either a dose-response interpretation (consistent supplementation produces a stronger within-person tilt) or a selection interpretation (members who log magnesium consistently are also more conscientious self-quantifiers, with cleaner within-person comparisons). Observational data cannot separate the two. The other notable gap is by sex.
Male members (N=386) showed an effect approximately 60 % larger than female members (N=1,272) on sleep score (d=0.53 vs 0.35), with a parallel pattern on REM sleep (4.5 min vs 3.5 min). The gap holds across age bands within each sex. We do not have a clean mechanistic explanation. Form differences were small. Generic magnesium and magnesium bisglycinate moved sleep score identically (d=0.38 in each cohort). The magnesium L-threonate sub-cohort (N=72) fell below the per-cell minimum and was not analysed.
The cohort design has the standard limitations of observational within-person work. Members who log magnesium in a sleep-focused application are a self-selected subset of the broader user base, with above-average baseline interest in sleep. Tag attribution depends on member behaviour, and there is no guarantee of dosing fidelity, timing relative to bedtime, or product authenticity. The most likely confound running counter to interpretation is reverse-direction logging: members may tag magnesium specifically on nights they expected, or had recently experienced, disrupted sleep, biasing the tagged-night side of the comparison toward worse-baseline nights. The remaining six contrasts move in the direction the mechanistic and clinical literature would predict, and the within-person sign-flip sensitivity check (random per-member sign permutation of the diff vector, three random seeds) collapses the cohort sleep-score difference from 1.88 to approximately 0.04 across replicates, which is the expected behaviour if the observed effect is signal rather than pairing artefact.
What this analysis adds is scale and within-person resolution. The paired n is two orders of magnitude larger than the largest published placebo-controlled magnesium-and-sleep trial. The result is consistent with the modest, real effect that the recent rigorous trials have been finding, while the rate-vs-variability split is the kind of distinction those trials are too small to make. The framing the analysis cannot make, and which only a randomised trial can, is causal. But the question of whether what magnesium is doing in clinical studies shows up at all in the field, in real members on real nights, has now been answered in the affirmative for sleep score and for overnight heart rate, and in the negative for overnight heart-rate variability.
- Chan V, Lo K. Efficacy of dietary supplements on improving sleep quality: a systematic review and meta-analysis. Postgraduate Medical Journal, 2021. PMID: 33441476.
- Gallagher C, Austin V, Dunlop KA, et al. Effects of supplementing zinc magnesium aspartate on sleep quality and submaximal weightlifting performance, following two consecutive nights of partial sleep deprivation. Nutrients, 2024. PMID: 38257144.
- Hausenblas HA, Lynch T, Hooper S, et al. Magnesium-L-threonate improves sleep quality and daytime functioning in adults with self-reported sleep problems: a randomized controlled trial. Sleep Medicine: X, 2024. PMID: 39252819.
- Schuster J, Cycelskij I, Lopresti A, Hahn A. Magnesium bisglycinate supplementation in healthy adults reporting poor sleep: a randomized, placebo-controlled trial. Nature and Science of Sleep, 2025. PMID: 40918053.
- Yeom JW, Cho CH. Herbal and natural supplements for improving sleep: a literature review. Psychiatry Investigation, 2024. PMID: 39086164.